 /GettyImages-1096547948-35b3799817ca4b2fa06888893ef4a348.jpg)
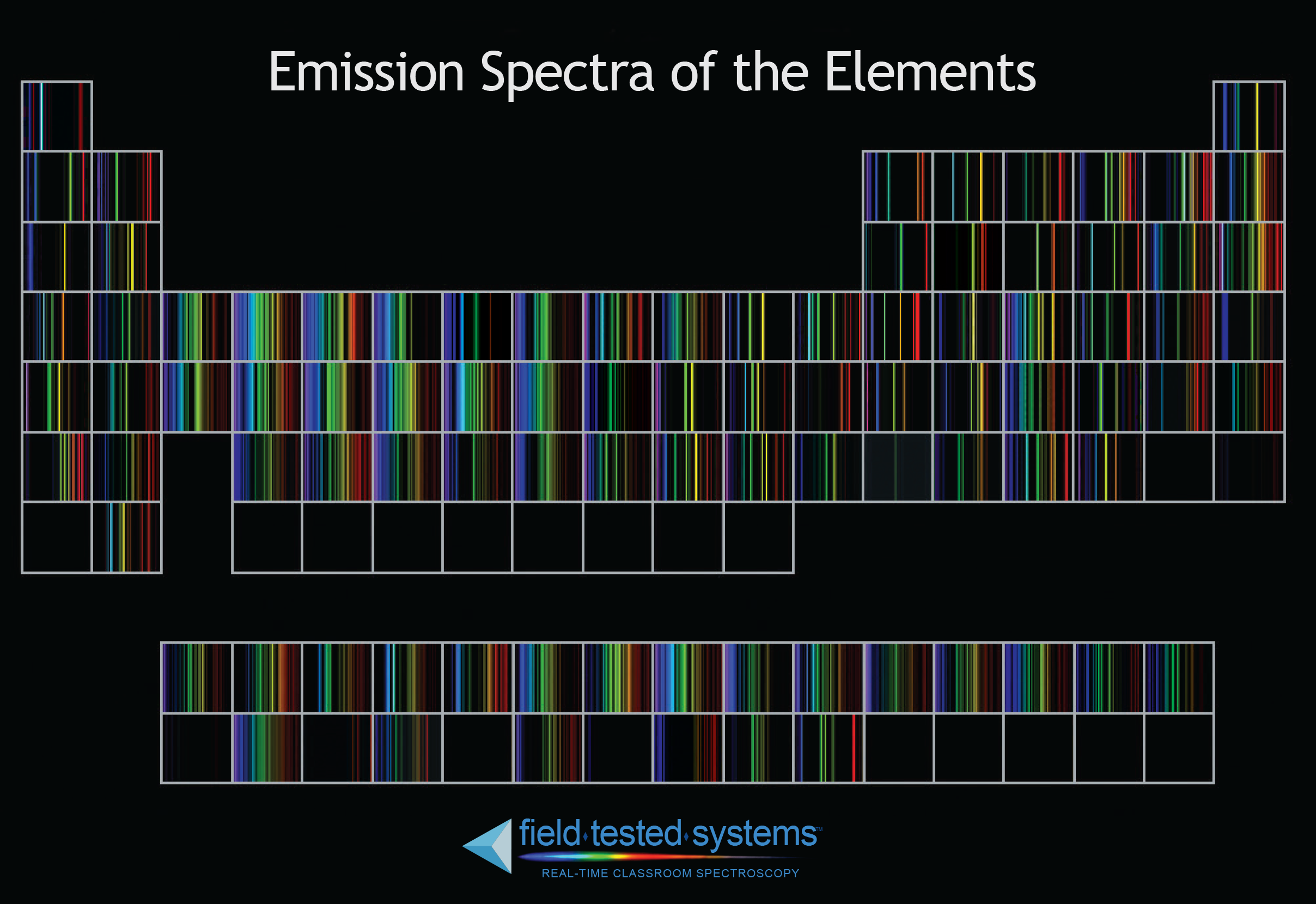
Because each element (and also each type of molecule) has its Only with the high energy orbits that are inaccessible at low temperature.Ĭomposition. Or have escaped altogether from their atoms, causing emission of specific lines associated The higher the temperature, the more electrons are in high energy orbits (can be more reliable than looking at the wavelength peak) That there is fundamental uncertainty in what will happen in the future (Figure from The EssentialĪstronomical sources has been a key to our understanding of the Universe because spectra Leading to a scientific confrontation with the philosophy of determinism: science shows We cannot predict precisely what they will do, Little dots, quantum mechanics tells us that they cannot be located accurately and are inįact more like fuzzy little fog clouds. Positive nucleus, emitting photons over a range of energiesĪlthough it is convenient to draw protons, neutrons, and electrons as Electrons over a range of energies can be captured by the Longer transitioning between two specific energy states, the atom can absorb a range ofĮnergies in this situation. Lavery, If even more energy is supplied to an electron, it canĮscape from the atom leaving the positively charged nucleus. In the gas are removed from the light that comes to us.Īn emission line spectrum is produced when photonsĪre emitted by gas that is thin enough to be transparent in the continuum. Rieke)Īn absorption line spectrum is produced when cool gas lies betweenĪ continuum source and us the specific wavelengths absorbed by the atoms Sources and gases involved (animation by G. Lines in a spectrum or absorption lines depending on the relationships of the the In astronomical situations, we may see either emission The electron falls back down to its original orbit.Įlectrons can also be raised to outer orbits when atoms collideĪ photon of the characteristic energy is emitted when the electron falls back to (The Amazing World of ElectronsĪ photon of the same energy is emitted when Photon depend on the energy difference between the orbits:Įnergy "hits" an atom, it can be absorbed and cause an electron to jump to an Lower energy orbit, energy is released in the form of photon. If an electron moves from an outer, higher energy orbit to an inner, Įlectron transitions between energy levels lead to absorption orĮmission of photons of specific energy corresponding to the energy level difference. The branch of physics called "quantum mechanics." (Figure by G. Levels in an atom because wave-particle duality means they interfere with The nucleus of the atom are held together by the "strong force", which isĬlearly much stronger than the electric one but works only over very small distances. This force attracts positive and negative electricĬharges, but repels like charges - two positives or two negatives. The electrons in an atom are held by the electric force, which is proportional toġ/r 2 just like gravity. With up to about 100 protons, giving 100 elements (figure by G. Hydrogen (1 proton) and helium (2 protons) are the simplest there are atoms Gases can have more complex emission- and absorption-line spectra,Īllowing us to learn a lot about their conditions.Įmission- and absorption-line spectra are produced by atoms (and molecules)Ītoms consist of nuclei made of protons and neutrons, and electronsĪround them. Is a particular type of continuous spectrum. Rutherford’s earlier model of the atom had also assumed that electrons moved in circular orbits around the nucleus and that the atom was held together by the electrostatic attraction between the positively charged nucleus and the negatively charged electron.Light at all the wavelengths. Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. In 1913, a Danish physicist, Niels Bohr (1885–1962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
